|
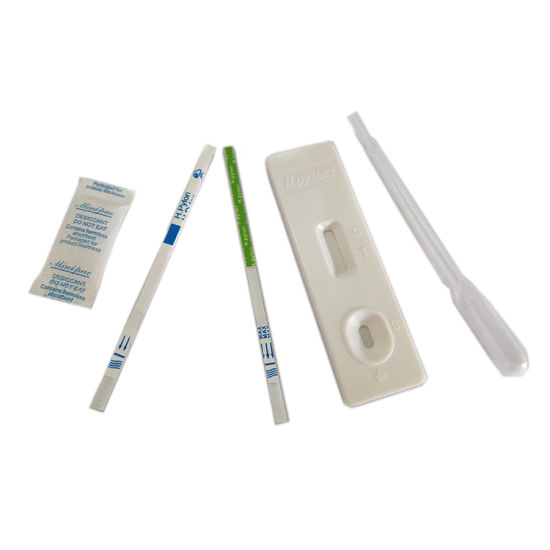
Format
|
strip, cassette
|
|
Specimen
|
Whole blood/serum/plasma/Feces |
|
Accuracy
|
Over 99%
|
|
Read time
|
10-15mins
|
|
Expiry date
|
24-36months
|
|
Certificate
|
CE, FSC,ISO13485
|
¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯
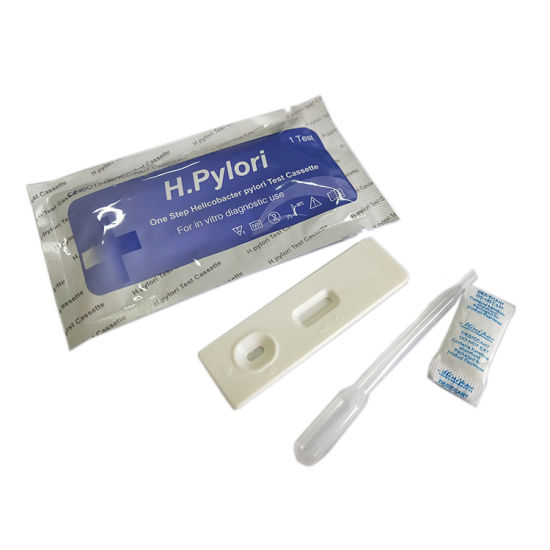
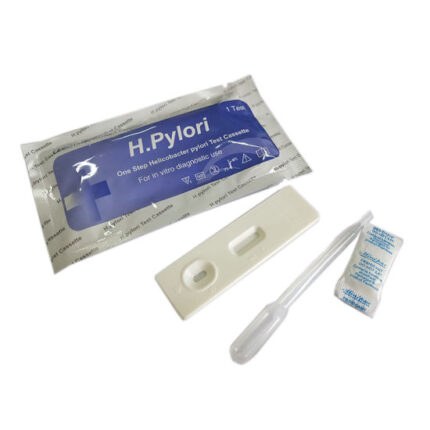
********* H.pylori Ab (Helicobacter pylori) test kit *********
Product Principle of H.pylori Ab test kit
The infection of Helicobacter pylori (H. pylori) is associated with a variety of gastro-intestinal diseases, such as stomach
ulcers, chronic active gastritis and gastrointestinal adenocarcinoma.
Antibodies to H. pylori are developed in individuals infected with H. pylori as a serological response.
Detecting specific antibodies to H. pylori can be used as a qualitative assay in the diagnosis of H. pylori infection, either
as an adjunct to endoscopy or as an alternative measure in symptomatic patients.
The H. Pylori Rapid Test is a chromatographic immunoassay (CIA) for the rapid determination of antibodies to H. pylori in
serum/plasma/whole blood specimens.
The test is to be used as an aid in the diagnosis of infection due to H. pylori.
|
Product type
|
strip, cassette
|
|
Usual size
|
Strip: 2.5mm or 3.0mm Cassette: 3.0 or 4.0mm
|
|
Specimen
|
Whole blood, serum/plasma |
|
Application period
|
Primary infection period or with symptoms |
| Type 1. H. pylori Ab test Strip Size: | 2.5mm 3.0mm |
| Type 2. H. pylori Ab test Cassette Size: | 3.0mm 4.0mm |
| Type 3. H. pylori Ag test cassette Size: | 3.0mm, 4.0mm |
| Storage | Indoor temperature of 2-30 degrees |
| Advantage | easy to use, fast to read result, accuracy and stable application, private usage |
¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯
|
Reagent
|
Control reagent kit
Positive Negative
|
Total
|
Sensitivity: Se=100% Specificity: Sp=100%
False positive rate =0%
False negative rate =0%
Total accordance rate = 100%
|
|
|
Positive
|
100
|
0
|
100
|
|
|
Negative
|
0
|
140
|
140
|
|
|
Total
|
100
|
140
|
240
|
|
Store test device unopened at 2°C-30°C. If stored at 2°C-8°C, ensure that the test device is brought to room
temperature before opening. Do not freeze the kit or expose the kit over 30°C.
Specimen of H. pylori Ab test Collection and Handling
For whole blood:
Collect whole blood into a collection tube by venipuncture. If specimens are not immediately tested, they should be
refrigerated at 2-80C. Freezing is recommended if the storage periods are more than 3 days. They should be brought to
room temperature before testing. Using the specimen after long-term storage of more than three days can cause
non-specific reaction. When stored at 2-8 0C, the whole blood sample should be used within three days.
For serum/plasma:
Blood should be collected aseptically into a clean tube without anticoagulants. Separate the serum or plasma from blood
as soon as possible to avoid hemolysis. Use clear non-hemolyzed specimens when possible.
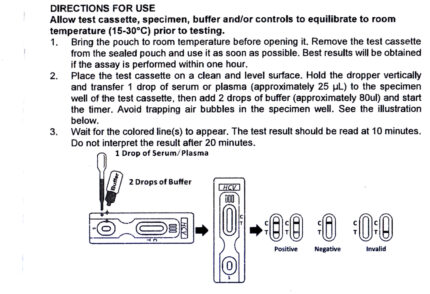
Procedure of H. pylori Ab test kit
> Step 1: When you are ready to begin testing, open the sealed pouch by tearing along the notch. Remove the test
from the pouch.
> Step 2: For Strip Test:
Immerse the strip into the container wtih the arrow end pointing towards the container.
Do not immerse past the MAX(maximum) line.
Take the strip out after 8-10 seconds and lay the strip flat on a clean, dry, nonabsorbent surface
(e.g., mouth of the serum container).
For Cassette Test:
Drip 2 or 3 drops of specimen into the sample window.
>Step 3 : Wait 10-15 minutes and read result. Do not read results after 30 15 minutes.
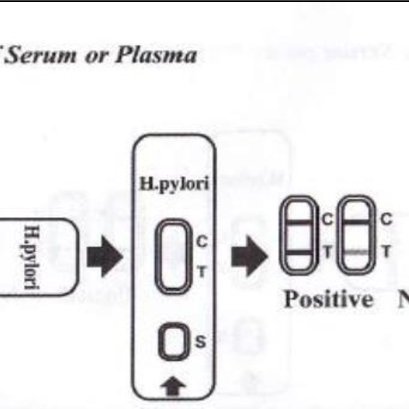
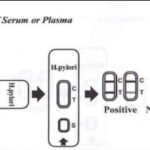
How to read the result of H. pylori test kit
>1. Negative: Only one pink band appears on test region.
>2. Positive: Two pink bands appear on test region.
>3. Invalid: If without colored band appears on test region, this is an indication of a possible error in
performing the test.
The test should be repeated using a new device.
 |
 |
WARNINGS AND PRECAUTIONS FOR Using H. pylori Ab test kit
≡1. Must use fresh specimen and avoid repetitive freezing, the result will be invalid
≡2. Use it before expiry date.
≡3. The package of kit should not be opened until it reaches the room temperature if it taken out from the refrigerator.
≡4. Old Serum can not be used. If the serum is thick, it can be used only after being separated.







![Polylab ]Plasticlabware](https://stinternationalbd.com/wp-content/uploads/2024/05/download.jpeg)













Reviews
There are no reviews yet.